AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog

Introduction of the complex combination of ECM proteins into liver tissue scaffolds has proven to drive altered functional responses from hepatocytes. Introducing this complexity into hepatocyte scaffolds can be achieved by stripping cells from dissected liver tissues and incorporating the decellularised liver ECM (dLECM) into scaffold structures. However, this does not capture the complexity present within native ECM which is composed of hundreds of different components categorized as Collagens, ECM glycoproteins, Proteoglycans, ECM regulators, ECM affiliated proteins and secreted factors. Hepatocytes rely on a dynamic relationship with a particular set of ECM proteins which governs the function of hepatocytes in‐vivo and facilitates communication between neighboring cells and the immune system.ĮCM proteins such as Collagen, Laminin and Fibronectin can be incorporated into electrospun polymer scaffolds, introducing cell‐matrix interactions that improve cell attachment and even direct cell function and differentiation. While these materials can provide a consistent structure with cell attachment motifs, these systems lack the specific biochemical niche that is present within the in‐vivo ECM. The bulk of many electrospun scaffolds is generally fabricated from biocompatible synthetic or natural polymers. Moreover, many studies have shown that the local mechanics of cell attachment substrates is proven to direct differentiation of both hepatocytes and stem cells. The morphology of electrospun fibers can be tailored to alter the local mechanical environment upon which cells attach and this has shown the affect the behavior and function of hepatocytes on electrospun polycaprolactone (PCL) scaffolds. This realistic biomechanical environment entails a simple fabrication process and research has proven its potential in the culture of hepatic cell types for in‐vitro models and regenerative cell therapy purposes. Progressions in 3D cell culture methods have delivered more realistic models of liver tissues in‐vitro, both structurally and functionally.Ĭulturing cells within a 3D matrix that recapitulates complex in‐vivo cell–cell and cell–matrix interactions allows for more accurate modeling of in‐vivo cell behaviors.Įlectrospun scaffolds provide a fibrous polymeric matrix like that of the in‐vivo extra‐cellular matrix (ECM) upon which cells can be attached in‐vitro. 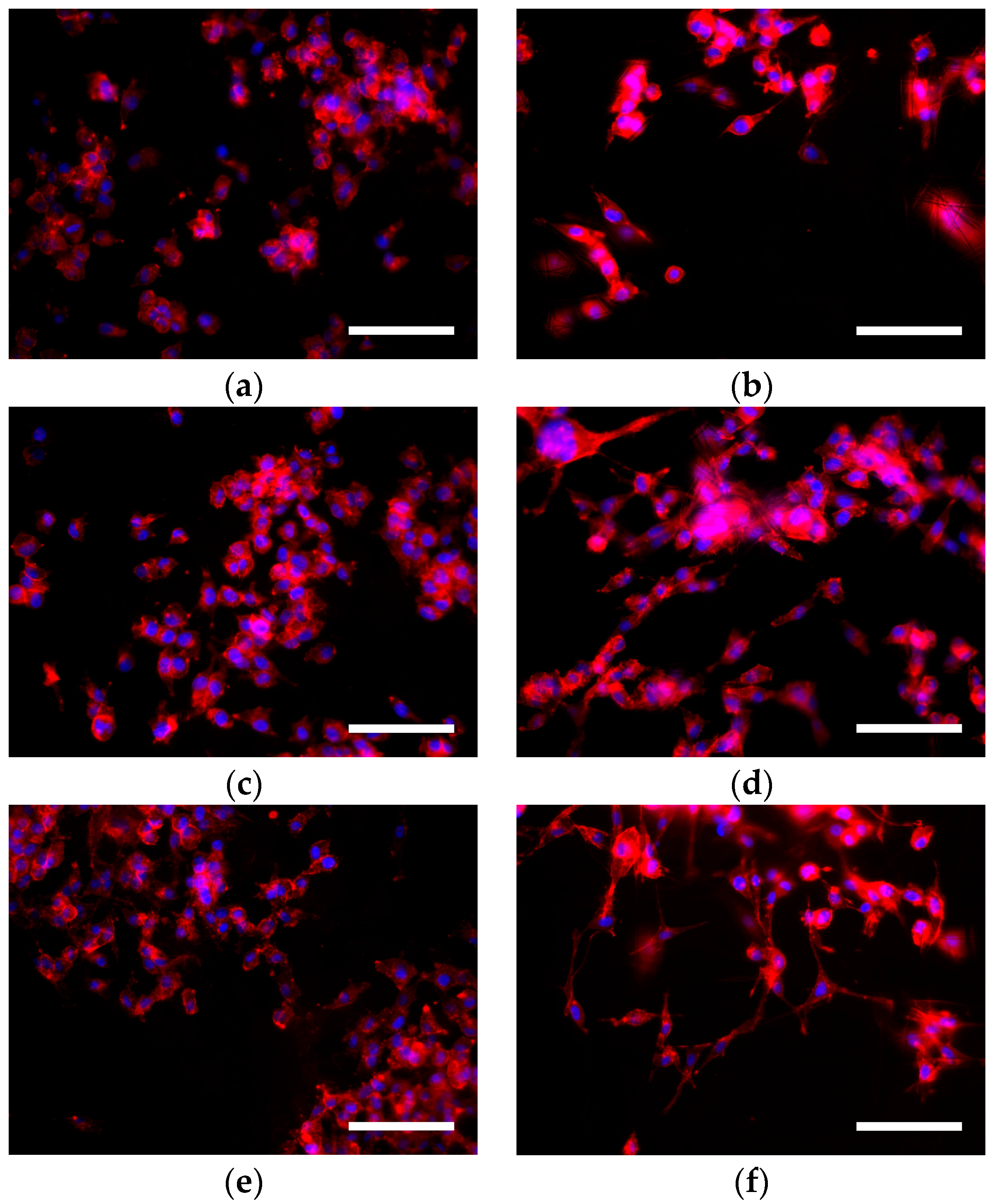
Thus, alternative strategies must be explored for the progression of drug development methodologies and other potential treatment methodologies for liver disease. This puts a quarter of the world at risk of fibrosis and hepatocellular carcinoma where currently no approved pharmaceutical treatment exists for any stage of liver disease and the eventuality of liver failure brings death without transplant liver availability. The burden of liver disease on people and health systems is increasing globally year‐on‐year and estimates now state that approximately 25% of the global population will be showing signs of onset or fully fledged NAFLD. This study confirms that rLECM can be utilized to modulate the bioactivity of electrospun PCL scaffolds and has the potential to produce electrospun scaffolds suitable for enhanced hepatocyte cultures and in‐vitro liver tissue models. Results showed significant increases in proliferative activity of HepG2 on rLECM containing scaffolds alongside maintained key gene expression. The hepatocellular carcinoma cell line, HepG2, was cultured upon the scaffolds for 14 days and were analyzed through cell viability assay, DNA quantification, albumin quantification, immunohistochemistry, and RT‐qPCR gene expression analysis. rLECM PCL scaffolds have been produced with 5 w/w% and 10 w/w% rLECM:PCL and were analyzed by SEM imaging, tensile mechanical analyses and FTIR spectroscopy. Herein, we present a methodology for incorporating whole organ decellularised rat liver ECM (rLECM) into polycaprolactone (PCL) electrospun scaffolds with the aim of producing biologically relevant liver tissue models. The development of pharmaceutical mitigation strategies is clearly necessary and methods to improve drug development processes are considered vital for this purpose. Organ transplantation is the only treatment for liver failure and with increasing cases of liver disease, transplant programs increasingly cannot provide timely transplant availability for all patients. Liver disease is expanding across the globe however, health‐care systems still lack approved pharmaceutical treatment strategies to mitigate potential liver failures.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
